Please allow ads on our site
Please log in to access this content. You will be redirected to the login page shortly.
LoginArrange the following compounds in increasing order of their property as indicated:
(i) Acetaldehyde, Acetone, DI-tert-butyl ketone, Methyl tert-butyl ketone (reactivity towards HCN).
(ii) CH_(3)CH_(2)CH(Br)COOH,CH_(3)CH(Br)CH_(2)COOH,(CH_(3))_(2)CHCOOH,CH_(3)CH_(2)CH_(2)COOH
(acid strength).
(iii) Benzoic acid, 4-Nitrobenzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength).
Class 12 Chemistry in Class 12 3 years ago
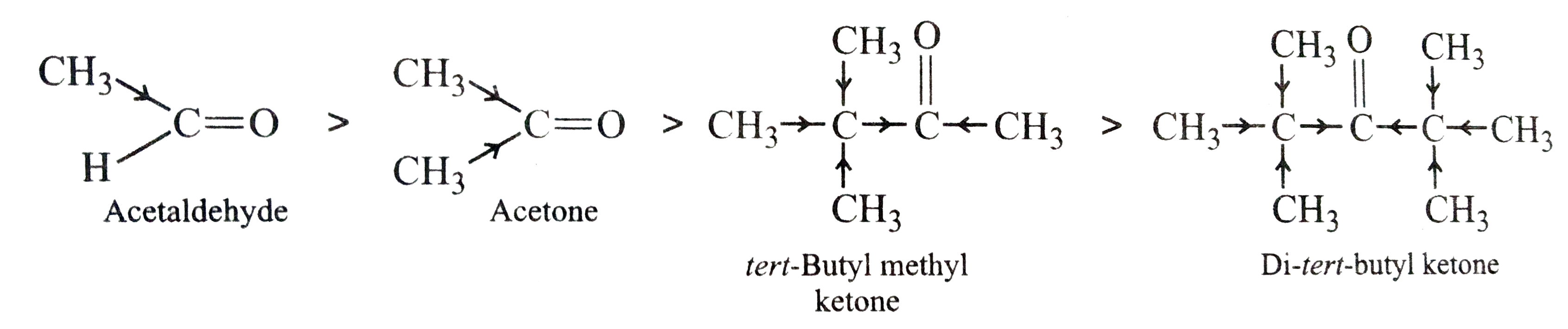
In other words, reactivity increases in the REVERSE order, i.e.,
Di-tert-butyl ketone lt tert-butyl methyl ketonelt acetonelt acetaldehyde.
(ii) we KNOWN that +I-effect decreases while -I-effect increases the acid strength of carboxylic acids.
Since +I-effect of isopropyl group is more tha the of n-propyl group, therefore, `(CH_(3))_(2)CHCOOH` is a weaker acid than `CH_(3)CH_(2)CH_(2)COOH`.
Further Since -I-effect decreases with distance, therefore, `CH_(3)CH_(2)CH(Br)COOH` is a stronger acid than `CH_(3)CH(Br)CH_(2)COOH`. thus, the overall acid strength increases in the order:
`CH_(3)-overset(CH_(3))overset(|)(C)H-COOH lt CH_(3)CH_(2)CH_(2)- COOH lt CH_(3)-UNDERSET(Br)underset(|)(C)H-CH_(2)-COOH lt CH_(3)-CH_(2)-underset(Br)underset(|)(C)H-COOH`
(iii) Since electron-donating GROUPS decrease the acid strength, therefore, 4-methoxybenzoic acid is a weaker acid than benzoic acid.
Further since electron-withdrawing groups inicrease the acid strength, therefore, both 4-nitrobenzoic acid and 3-4-dinitrobenzoic acids are stronger acids than benzoic acid further due to the presence of an additional `NO_(2)` group at m-position w.r.t. `COOH` group, 3,4-dinitrobenzoic acid is a little stronger acid than 4-nitrobenzoic acid. thus, the overall acid strength increases in the order:
4-Methoxybenzoic acidltBenzoic acidlt4-Nitrobenzoic acidlt3,4-Dinitrobenzoic acid.
Posted on 04 Dec 2021, this text provides information on Class 12 related to Chemistry in Class 12. Please note that while accuracy is prioritized, the data presented might not be entirely correct or up-to-date. This information is offered for general knowledge and informational purposes only, and should not be considered as a substitute for professional advice.
Take Quiz To Earn Credits!
Turn Your Knowledge into Earnings.

No matter what stage you're at in your education or career, TuteeHub will help you reach the next level that you're aiming for. Simply,Choose a subject/topic and get started in self-paced practice sessions to improve your knowledge and scores.

